Gas Stove Ignition Coal Stove Frame Kitchen Hardware Accessories Iron Stove Frame Kitchen Accessories Can Be Customized - China Cast Iron and Grate Burner price | Made-in-China.com

Scientific amusements . Fig. 78.—Iron Filings burning in a Jet of Air. mixture. When the air has been withdrawn the hydrogen.gas can be lighted at the extremity of the tube. Hydrogen is

How the Future of Greenhouse Gas Emissions Produced by Jet Fuel May be Changing due to an Iron Catalyst

IMEISH Gas Stove 8000 W Cast Iron Burner with Gas Cooking Ring Large GPL Stove Outdoor Frame Iron Portable Stove with Fire Control : Amazon.com.au: Everything Else

Flow sheet over the steelmaking processes. Steel can be produced from... | Download Scientific Diagram

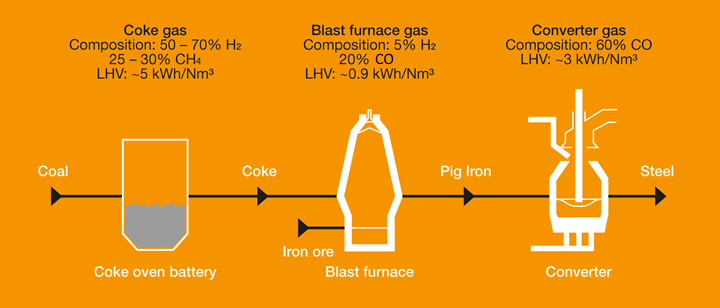
Chemical engineering . iron and steel works forsupplying fuel to gas engines when the waste blast furnacegases arc not adequate for providing sufficient power, butat present high power gas engines utilize these

Deoxidation Electrolysis of Hematite in Alkaline Solution: Impact of Cell Configuration and Process Parameters on Reduction Efficiency - Fayaz - ChemElectroChem - Wiley Online Library

I cook on gas and the flames seem to burn the bottom of my pans. "Seem" because although you can see the bare iron, the seasoning seems still to be there. How
What causes the colours that form on a clean iron or steel surface after it has been heated and cooled for tempering? - Quora